Ask anyone who has been through the process of finding the right psychiatric medication, and you will likely hear a version of the same story. The first medication did not work at all. The second caused side effects that were worse than the symptoms it was supposed to treat. The third helped somewhat, but not quite enough. By the time the right medication at the right dose was finally found—if it was found—weeks or months had passed, the person had suffered unnecessarily, and their confidence in the treatment process had been badly shaken. This experience is so common that many people simply give up. Precision psychiatry is determined to change it.
At the heart of precision psychiatry is a simple but powerful observation: people are not biologically identical, and the way their bodies process medications varies enormously from one person to the next. What works beautifully for one patient may be completely ineffective for another, and what is well tolerated by one person may cause serious side effects in someone else. For decades, this variability was treated as an unfortunate reality of psychiatric practice—trial and error was simply how it was done. Pharmacogenomics, the study of how genes influence drug response, has begun to replace that resignation with something more useful: information.
Note: Setting adhd appointment is the first step towards managing psychological conditions.
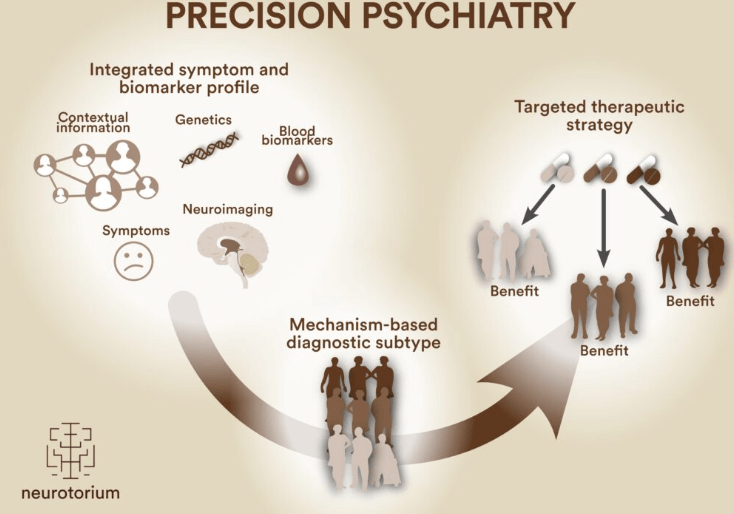
The genetic factors most relevant to medication response involve the liver enzymes responsible for metabolizing drugs. The most studied of these are the cytochrome P450 enzymes, particularly CYP2D6 and CYP2C19. Genes encoding these enzymes vary between individuals in ways that have significant consequences for how drugs behave in the body. Some people are “poor metabolizers”—their enzymes work slowly, meaning that standard doses of certain medications accumulate in their system to levels that produce toxicity or intolerable side effects. Others are “rapid metabolizers”—their enzymes work so efficiently that standard doses are broken down before they can have any effect, leaving the patient apparently unresponsive to medications that would work perfectly well at a higher dose.
A simple cheek swab or blood test can now identify which metabolizer category a patient falls into for many of the most commonly prescribed psychiatric medications. Armed with this information, a clinician can make much more informed decisions about which drugs to choose and at what dose to start. Instead of beginning with the medication that works for the statistical average and adjusting from there, the treatment can be tailored to the individual’s actual biology from the start.
Pharmacogenomic testing has shown particular utility in guiding the use of antidepressants. Many SSRIs and SNRIs—the first-line medications for depression and anxiety—are metabolized by CYP2D6 or CYP2C19. Knowing a patient’s metabolizer status can help explain why a person has repeatedly failed standard doses of multiple medications in the same drug class, or why they have experienced unusually severe side effects. For antipsychotic medications, where side effects can be particularly burdensome and treatment adherence is a persistent challenge, having genetic guidance about likely tolerability can make a meaningful difference in whether a patient stays on their medication or abandons it.
Beyond enzyme genetics, researchers are exploring other biological markers that may predict treatment response. Blood-based inflammatory markers have attracted attention because of growing evidence that a subset of depression is driven in part by chronic low-grade inflammation rather than by the serotonin deficiency that older theories emphasized. Patients with elevated inflammatory markers may respond better to treatments that address inflammation than to conventional antidepressants. Neuroimaging is being used in research settings to identify patterns of brain activity that predict which type of therapy—cognitive behavioral, interpersonal, or psychodynamic—a particular patient is most likely to benefit from.
Note: AI will continue to grow in the use and help for psychological conditions and the technology continues to get better and better.
The integration of these different biological data streams into what researchers call “precision psychiatry” reflects a broader shift in how the field thinks about mental illness. Rather than treating “depression” or “anxiety” as single, uniform conditions, this approach recognizes that these diagnostic categories likely contain many biologically distinct subtypes requiring different treatments. Just as oncology has moved from treating “cancer” generically to targeting specific mutations in specific tumor types, psychiatry is moving toward matching treatments to the biological reality of the individual patient.
This is not a fully realized vision yet. The evidence base for pharmacogenomic testing, while growing, still has limitations. Not all relevant genes have been identified. The predictive power of current tests, while useful, is not absolute—other factors including environment, life history, and co-occurring conditions influence treatment response in ways that genetics alone cannot capture. Insurance coverage for genomic testing remains inconsistent, creating equity concerns about who has access to this kind of individualized care.
But even in its current, imperfect form, precision psychiatry represents a meaningful improvement over what came before. Every avoided failed medication trial means weeks less suffering. Every accurately predicted side effect that never happens means a patient who stays in treatment rather than dropping out in frustration. The days of treating psychiatric medication as a guessing game are not fully over, but they are numbered—and that is good news for everyone who has ever felt like a guinea pig in their own care.